Abstract

Background: Multiple myeloma (MM) is characterized by dramatic biological and clinical heterogeneity. Prognostic biomarkers including genomic alterations and bone marrow (BM) minimal residual disease can resolve some of this heterogeneity; however, these rely on invasive tumor sampling which limits their utility. Here we develop and apply plasma cell-free DNA liquid biopsies via Cancer Personalized Profiling by Deep Sequencing (CAPP-Seq) to characterize tumors and quantify treatment response in patients with MM and explore its utility during anti-BCMA CAR T-cell therapy.
Methods: We developed a 480 kb MM-specific CAPP-Seq panel, which covers 86 recurrently mutated coding genes as well as other recurrently mutated regions, for disease detection and quantification from circulating tumor DNA (ctDNA). Using this hybrid-capture panel, we profiled 214 plasma, germline and tumor DNA samples from 35 patients including 15 patients undergoing idecabtagene vicleucel (ide-cel) therapy. Samples were sequenced to ultra-high unique molecular depth as previously described (Newman et al, Nat Biotechnol 2016) to quantify ctDNA. Copy number variants (CNVs) were identified using our recently developed method, CANARy (Chabon et al, Nature 2020). Tumor variants and ctDNA levels were identified prior to and following therapy to assess ctDNA dynamics, and were correlated with clinical indices and patient outcomes.
Results: We applied CAPP-Seq to samples from 35 patients with newly diagnosed (ND, n=10) or relapsed/refractory (RR, n=25) plasma cell disorders. A median of 87 single-nucleotide variants (SNVs, range 2-283) were detected per case. Among these, a median of 5 mutations/patient were in coding genes such as KRAS (25% of patients), TP53 (25%) and NRAS (19%). Most other SNVs occurred in genomic regions targeted by activation induced deaminase (AID), such as IGH, IGK and IGL. To validate tumor genotyping from plasma, we compared SNVs observed in plasma to paired BM samples in 18 cases. Here, we observed 84% (594/703) of all mutations identified in BM were also present in plasma, including 90% (26/29) of all coding alterations. Interestingly, we detected an additional 259 SNVs in the plasma that were not present in the BM, including KRAS, NRAS, and FAM46C mutations, suggesting ctDNA may better capture spatial tumor heterogeneity.
We next utilized CAPP-Seq to quantify disease burden prior to treatment. We detected ctDNA in 97% (31/32) of pretreatment plasma with a median allele frequency of 0.91% (range 0.006 to 36%) and 3.82 log haploid genome equivalents per mL of plasma (hGE/mL). Levels of ctDNA were higher in RRMM compared to NDMM patients (4.26 vs 3.08 log hGE/mL, p=0.02) and correlated with clinical risk factors including R-ISS (stage 1: 2.83 vs stage 2/3: 4.18 log hGE/mL, p=0.01) as well as disease burden measurements including M-spike (rho=0.53, p=0.02) and free light chain ratio (rho=0.40, p=0.03). The fold change in ctDNA level at a median of 36 days from treatment initiation correlated with clinical response (median 0.060 for non-progressors vs 7.0 for progressors, p=0.04).
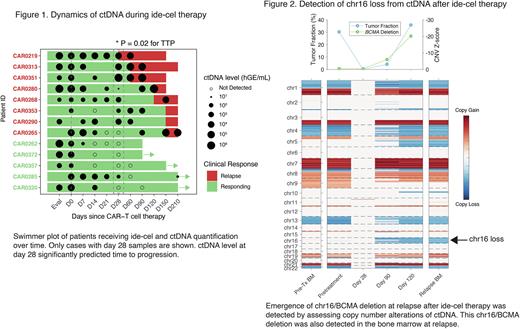
Further, we applied CAPP-Seq to serial samples from 15 patients with RRMM receiving ide-cel. Notably, patients with longer response (>90 days) had significantly lower ctDNA levels at day 28 compared to those with early (<90 days) progression (0.6 vs 4.1 log hGE/mL; p=0.009, Fig 1). Lower ctDNA levels at day 28 predicted longer time to progression (proportional hazard model, p=0.02, HR=1.6). Finally, we assessed clonal evolution upon relapse after ide-cel. In multiple patients we observed emergence of new CNVs in ctDNA, including a case with an emergent loss of chr16, where TNFRSF17 (BCMA) resides (Fig 2). This copy number loss was observed 36 days prior to clinical relapse, was validated through WGS of the relapsed tumor, and resulted in loss of BCMA by immunohistochemistry.
Conclusions: Assessment of ctDNA has significant potential utility in MM, as it allows for mutational genotyping, quantification of disease burden, and identification of tumor clonal evolution. Lower levels of ctDNA during treatment and its clearance are associated with improved outcomes, while detection of emergent alterations after targeted therapies may inform subsequent treatment strategies. Use of ctDNA may improve response and relapse prediction and reveal mechanisms of therapy resistance.
*SS and DMK are co-senior authors
Disclosures
Khodadoust:CRISPR Therapeutics: Research Funding; Nutcracker Therapeutics: Research Funding; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees; Myeloid Therapeutics: Membership on an entity's Board of Directors or advisory committees. Miklos:Allogene: Research Funding; Novartis: Consultancy; Pharmacyclics: Patents & Royalties: cGVHD Ibrutinib patent ; Kite, a Gilead Company: Research Funding; Janssen: Consultancy, Honoraria; Adaptive Biotech: Consultancy; Bristol Meyers Squibb: Consultancy; Fosun Kite: Consultancy, Honoraria. Arai:Kadmon: Membership on an entity's Board of Directors or advisory committees. Liedtke:GSK: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Natera: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncopeptides: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Gilead: Research Funding; Caelum: Research Funding; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Alnylam: Membership on an entity's Board of Directors or advisory committees; Allogene: Research Funding; Adaptive: Membership on an entity's Board of Directors or advisory committees; Kite: Membership on an entity's Board of Directors or advisory committees; Kura Oncology: Membership on an entity's Board of Directors or advisory committees; Seagen Inc.: Research Funding. Sidana:Prothena: Honoraria; Oncopeptides: Consultancy; Sanofi: Consultancy; Janssen: Consultancy, Research Funding; Magenta Therapeutics: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; Allogene: Research Funding. Kurtz:Adaptive Biotechnologies: Consultancy; Roche: Consultancy; Genentech: Consultancy; Foresight Diagnostics: Consultancy, Current equity holder in private company, Patents & Royalties.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract
Asterisk with author names denotes non-ASH members.